Fortoss Vital: Can This Be Used for a Patient with a Severe Buccal Concavity Over his Upper Central?
Dr. G. asks:
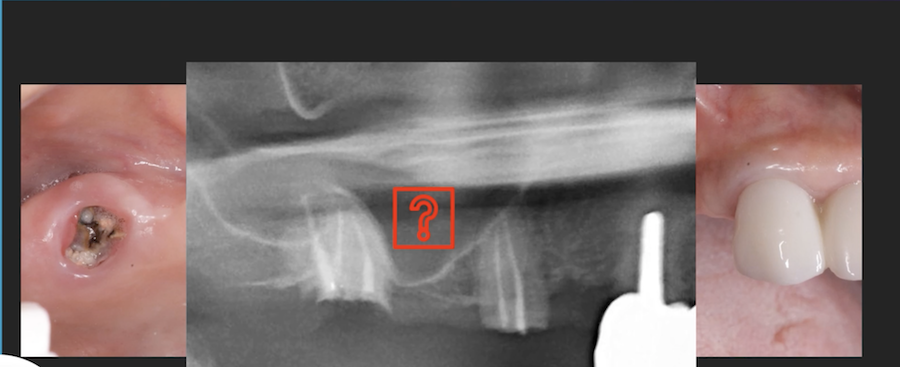
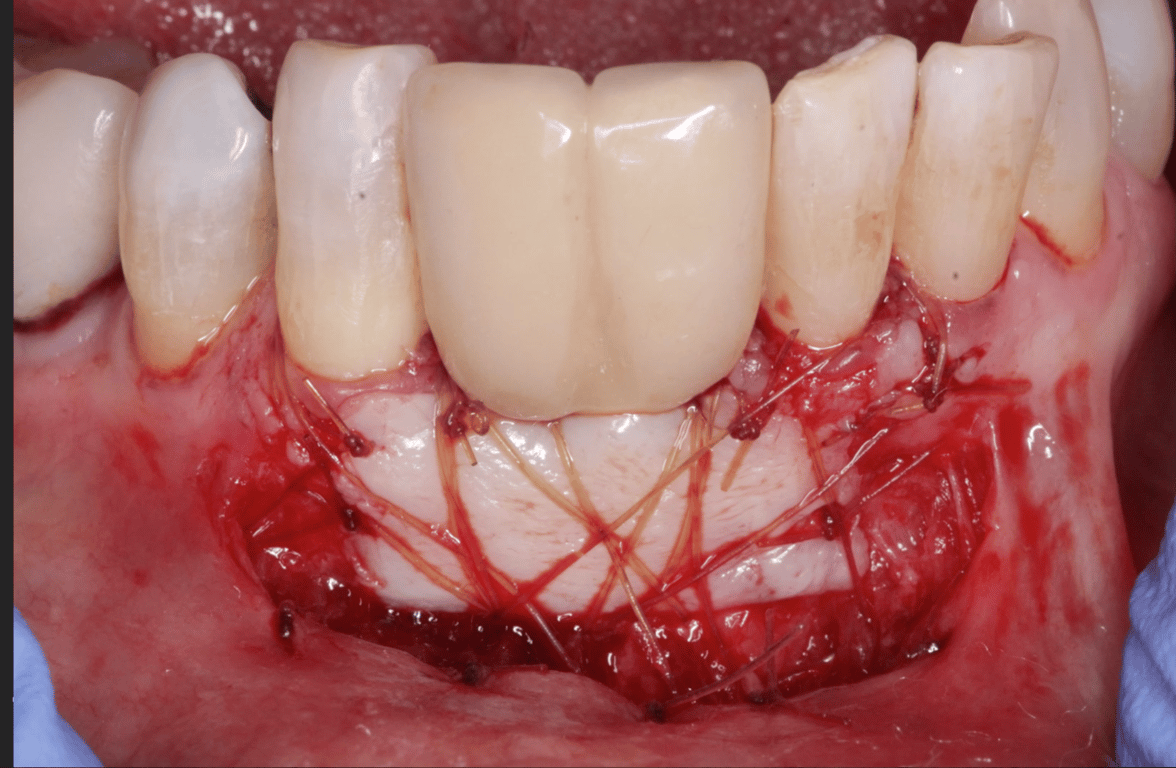
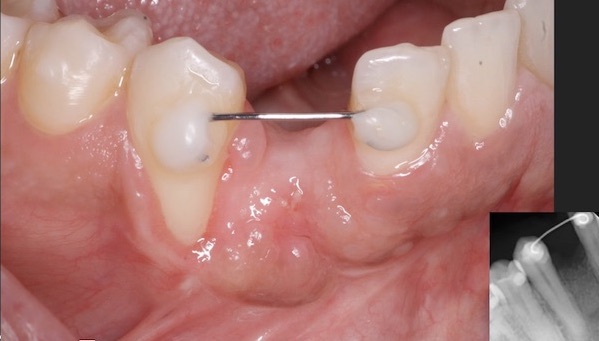
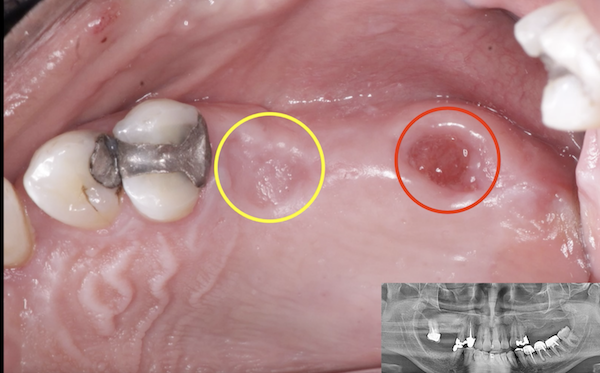
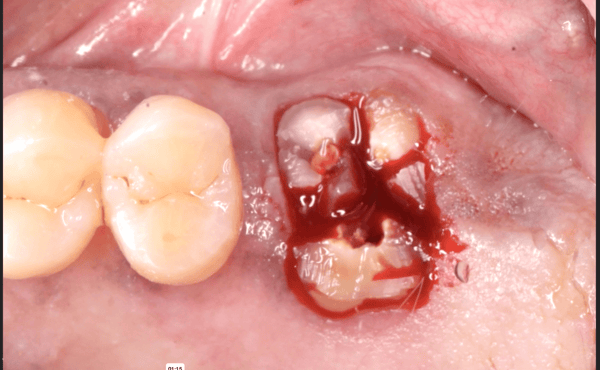
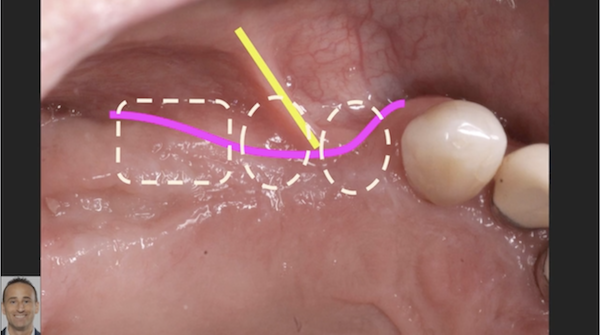
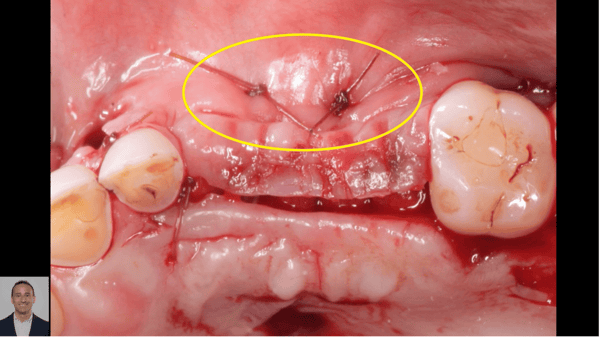
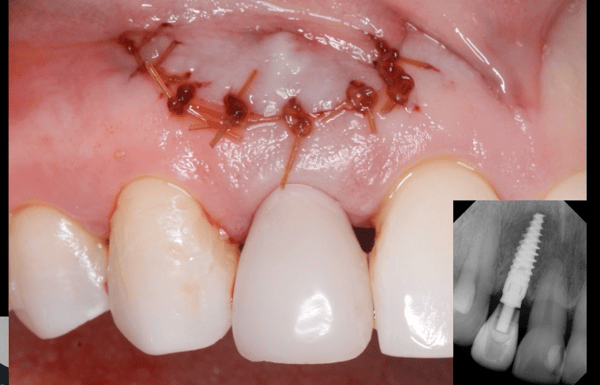
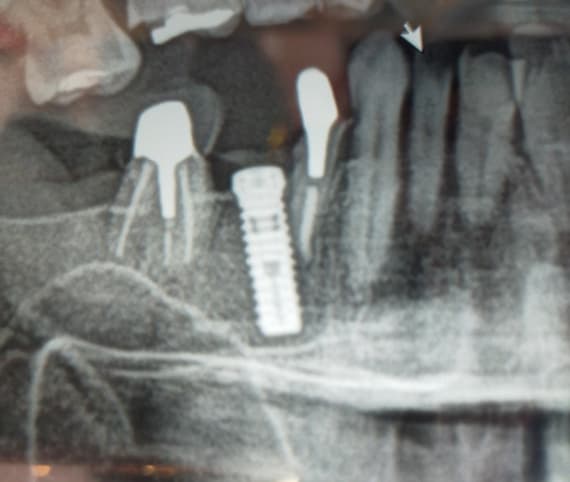
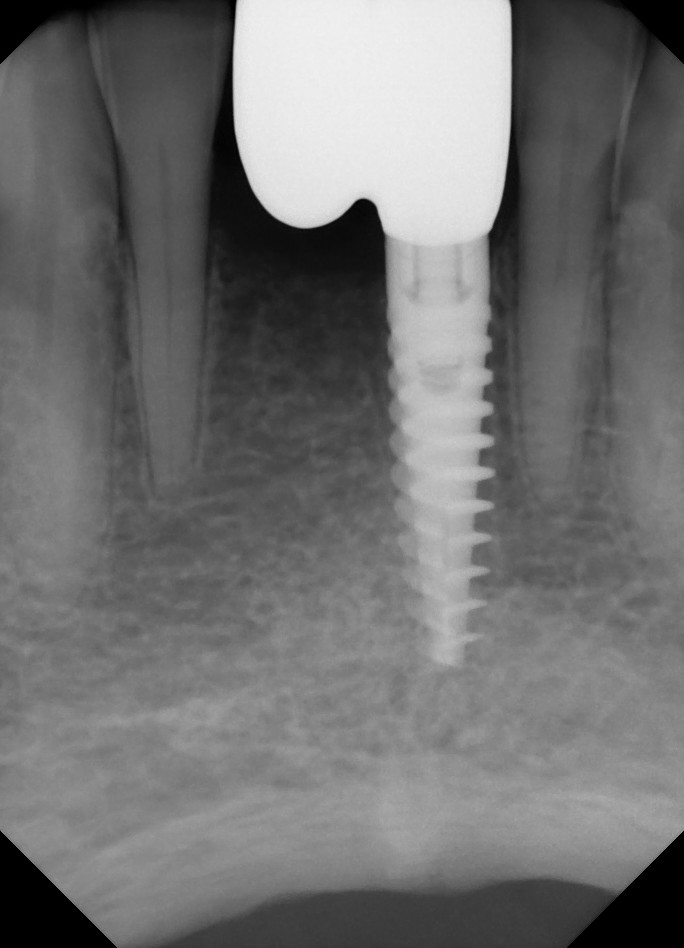
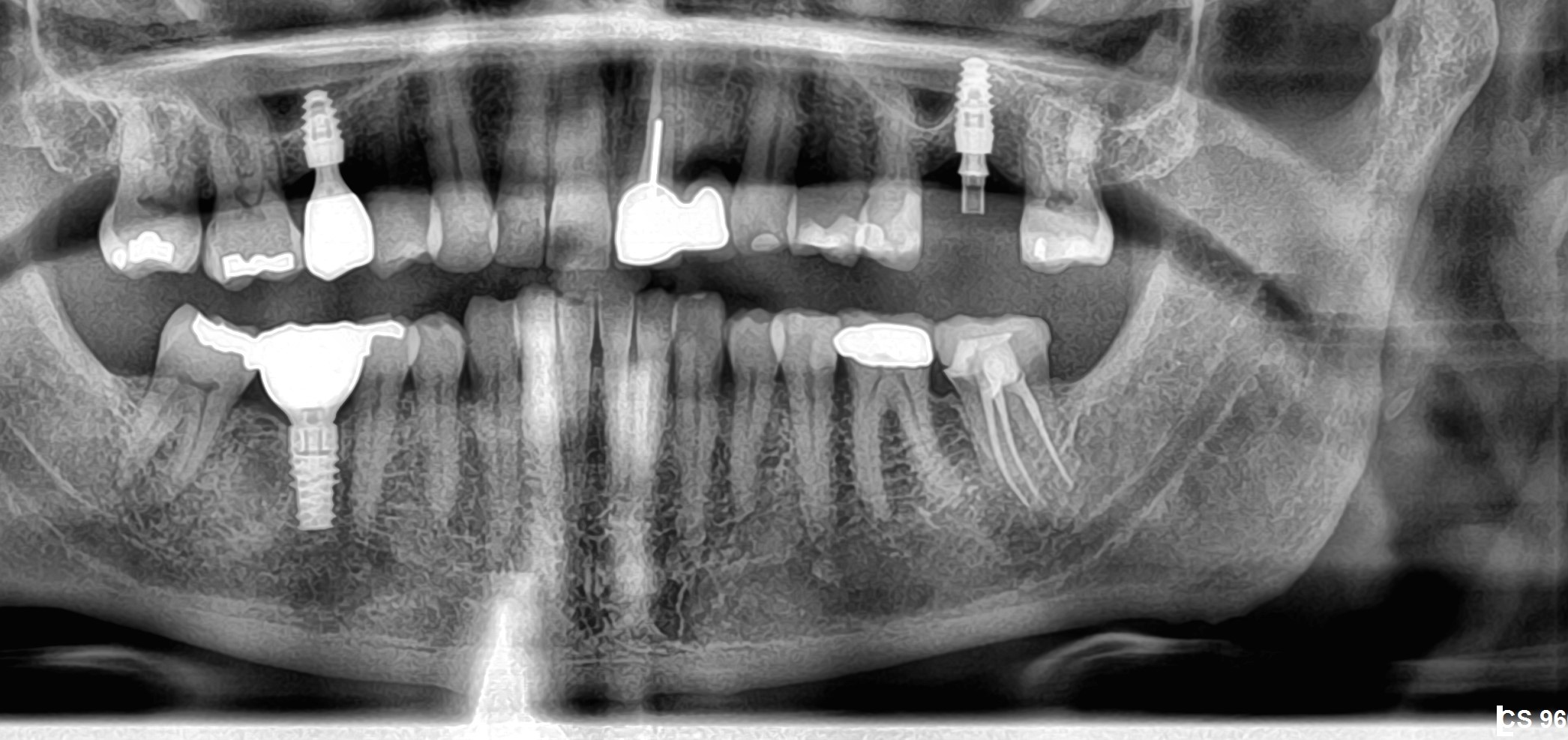
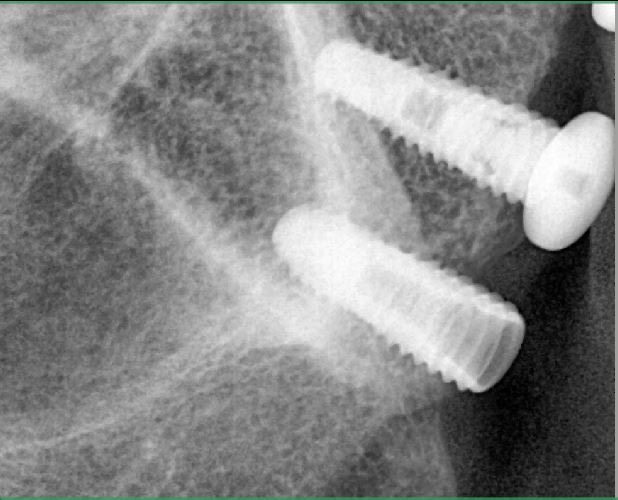
I have to place an implant in the #8 area [maxillary right central incisor; 11]. The patient has a pronounced concavity on the buccal that seriously compromises the available buccolingual bone volume. The buccal defect would best be corrected by a block graft. I was wondering if I could use the synthetic bone graft material [alloplast] Fortoss Vital. [The manufacturer describes this material as “… bi-phasic calcium composite bone graft material having a negative surface charge which attracts the key proteins and cells for rapid osteogenesis and bone repair. Fortoss Vital is fully resorbed by osteoclastic processes and replaced by bone…â€]. I would prefer to use a synthetic graft if possible. I would like to be able to re-enter the graft at 6 months to place the implant. Is the resulting bone formation derived from such a synthetic material, like Fortoss Vital, sufficient enough for implant placement or is a bone block inevitable?