Glidewell Dental Releases 3.2-Mm-Diameter Inclusive Tapered Implant
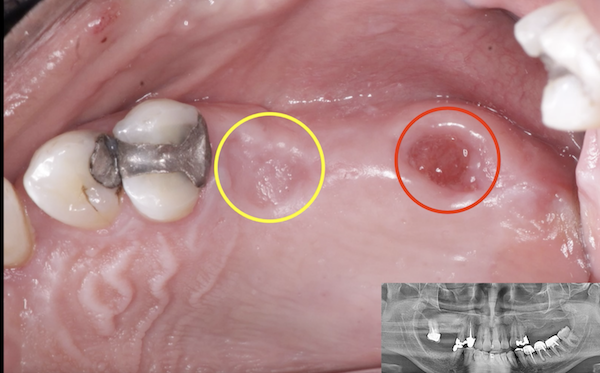
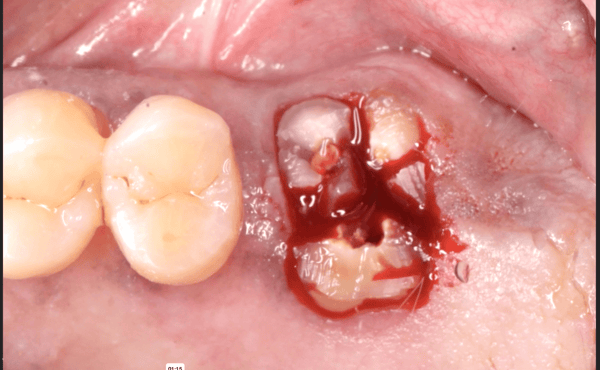
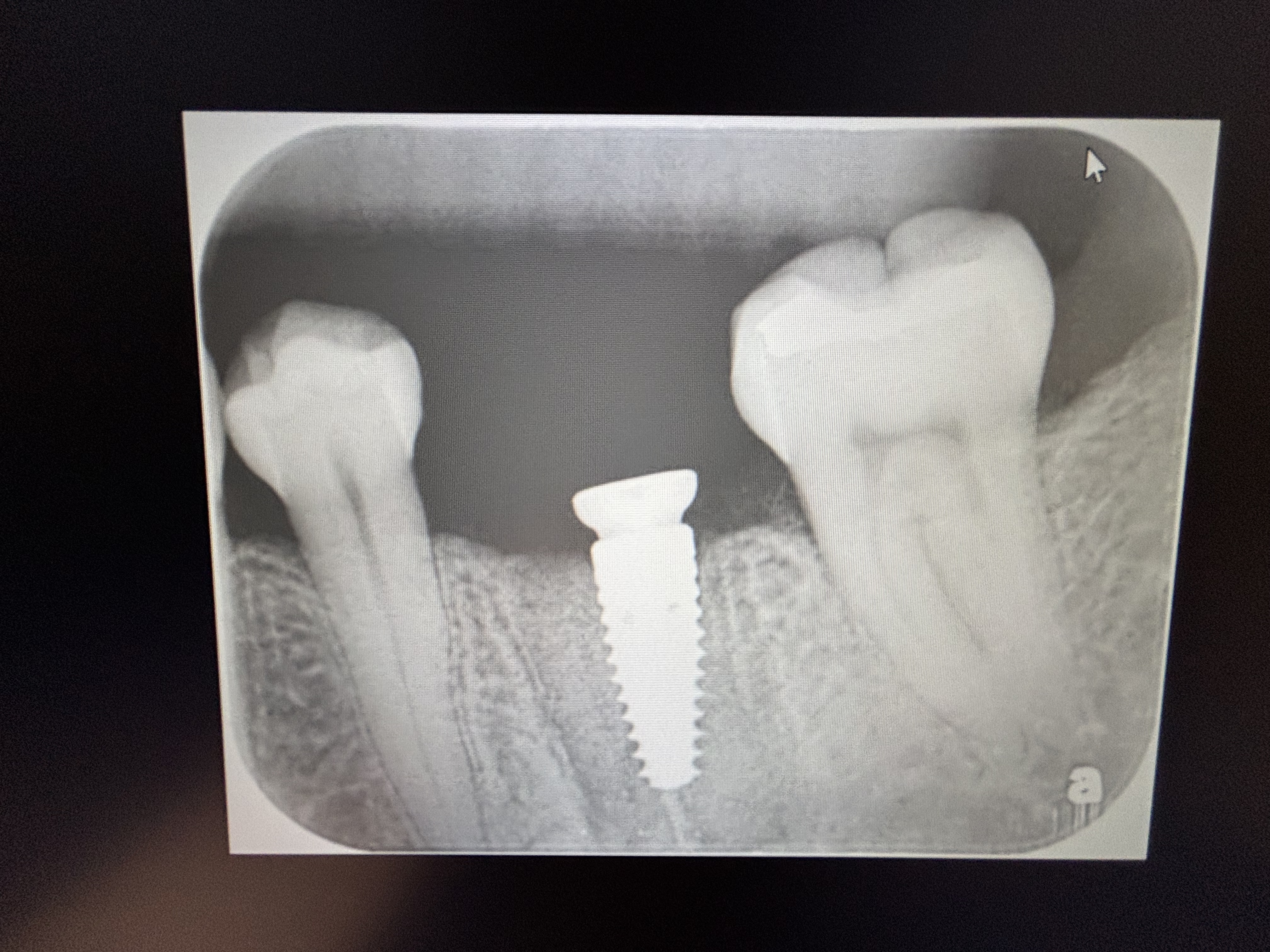
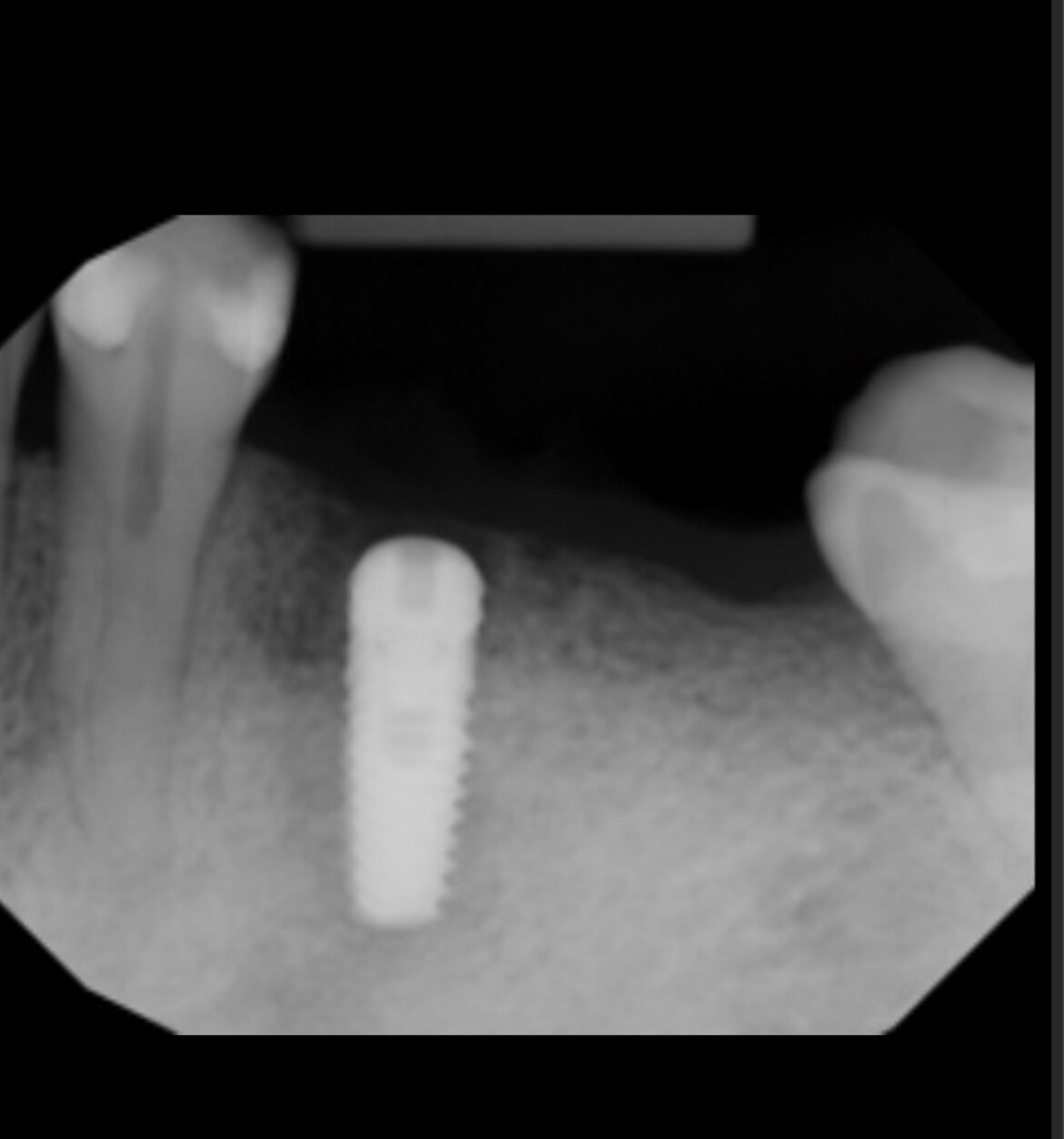
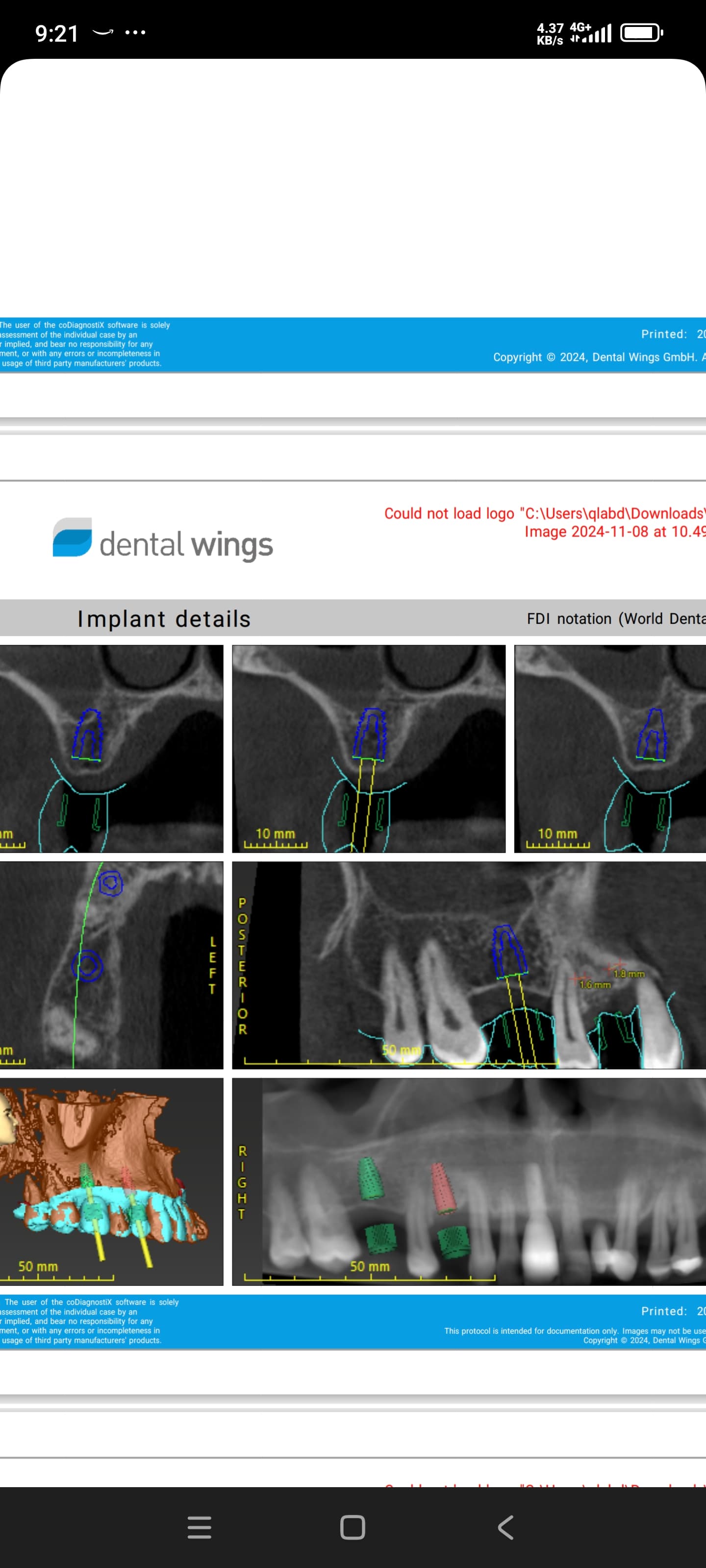
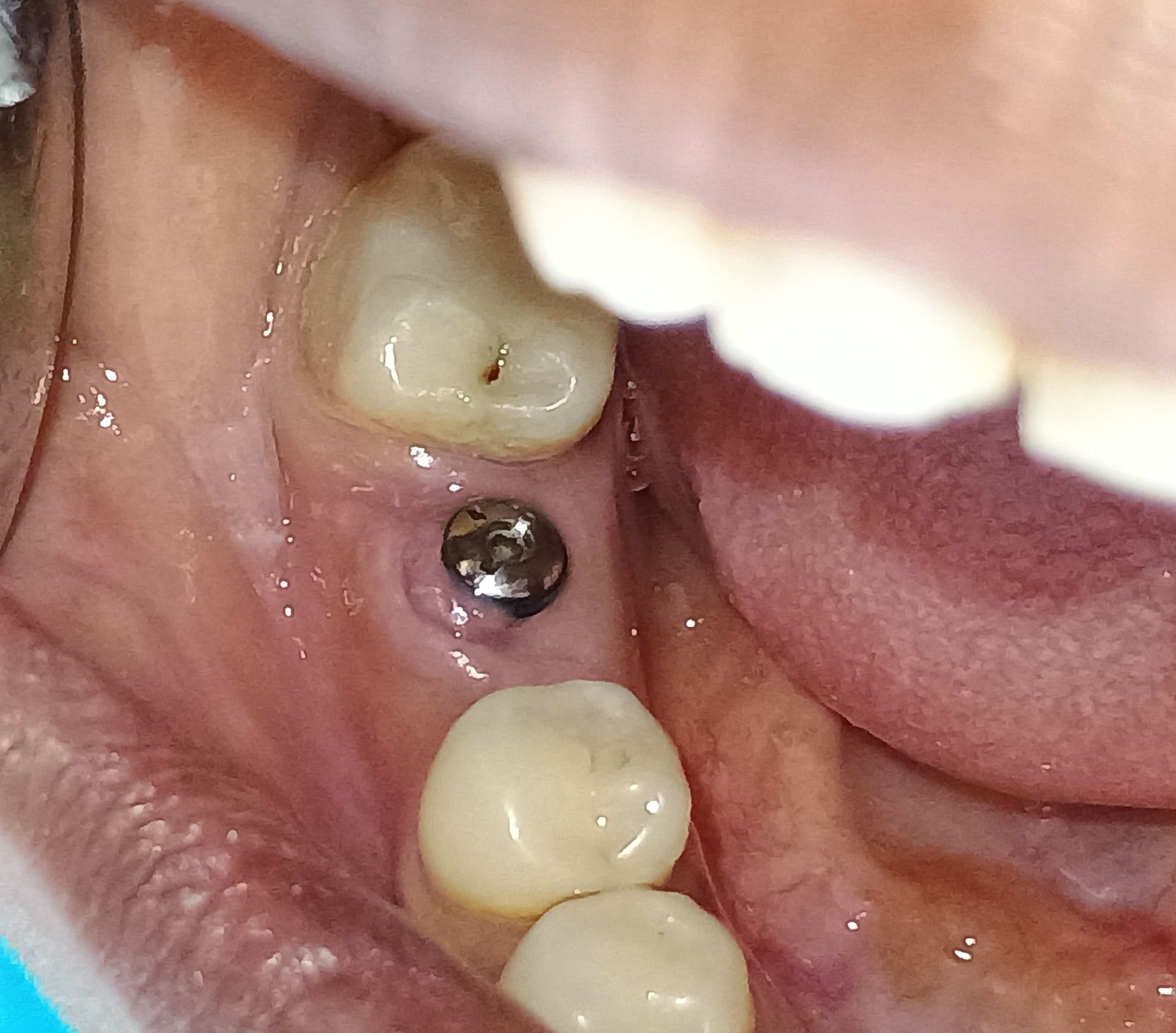
Glidewell Dental, industry-leading provider of dental products, implant solutions and lab services, announced today that the Inclusive® Tapered Implant is now available in a 3.2 mm diameter. In a continuation of the company™s commitment to provide practitioners with the tools they need to achieve predictable results in a wide variety of clinical situations, the new implant size incorporates a narrow-diameter apex and was designed to ease positioning in areas of limited anatomical space, such as upper lateral incisors, lower incisors and narrow ridges. With the latest addition, the Inclusive Tapered Implant is now available in 3.2 mm, 3.7 mm, 4.2 mm, 4.7 mm and 5.2 mm diameters.
The new 3.2-mm-diameter implant includes a 3.0 mm prosthetic platform, a design feature engineered to increase soft-tissue thickness at the abutment-implant connection, which can facilitate crestal bone preservation. Featuring an industry-standard, internally hexed conical connection, the implant is compatible with popular prosthetic components and instrumentation. The implant™s deep conical interface encourages lateral stability, while its coronal micro-threads increase the surface area available for bone-to-implant contact at the crest of the ridge. The tapered body of the implant and buttress threads are designed to engage and gently compress the bone, increasing primary stability and aiding the osseointegration process.
The addition of the 3.2 mm implant is accompanied by the release of an all-new surgical kit, which has been redesigned for greater simplicity, durability and ease of use, and expanded to accommodate the full range of Inclusive Tapered Implants.
Inclusive Tapered Implants have performed well in clinical studies, are machined from high-strength titanium alloy and include a surface that has been treated with resorbable blast media (RBM), a process that has been shown to promote bone development on the implant. For more information on the Inclusive Tapered Implant, call 800-407-3379 or visit inclusivedental.com.